Jordi Marti's Homepage
Computational Physics research group (Left to right: Edgar Alvarez-Galera, Yanhong Ge, Huixia Lu, Jordi Martí, Zheyao Hu, Arnau Prat)
Contact Details
| Name |
Prof. Jordi Marti |
|
| Address |
Department of Physics, Polytechnic University of Catalonia-Barcelona Tech, B5-209, Northern Campus, Jordi Girona 1-3, 08034 Barcelona, Catalonia, Spain |
|
| Telephone FAX |
+34 934017184 +34 934017100 |
|
| jordi.marti |

Research Interests
My main interest concerns the study of Condensed Matter Physics and Statistical Mechanics, focussing my work on the liquid phase. I spend most of my research activity working in theory and applications to the structure and dynamics of simple and complex liquids, with special attention paid to water, alcohols and aqueous electrolyte solutions. I have performed computer simulations with several methods: Molecular Dynamics, Monte Carlo, Empirical Valence Bond, Transition Path Sampling and Metadynamics, among others. In recent years, I have participated in the applications of computer simulation to biological systems, such as the study of oncogenes and the design of viable drugs for cancer treatment and on the interactions of proteins and small-molecules with biomembranes. I'm also working on proton transfer in aqueous environments and under restricted geometries, on the study of helium nucleation inside the lead-lithium blankets of nuclear fusion reactors and on the computation of partition functions in condensed matter systems using advanced tools such as machine learning and importance samping techniques.
Selected Topics
A. Liquid water and aqueous solutions
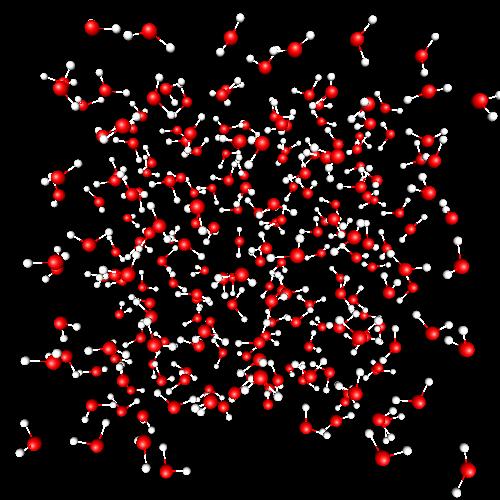
Liquid and supercritical water |
 |
|
A tetrahedral network of water molecules is the main structural trend, due to the existence of four hydrogen-bonds per molecule (on average). The lifetime of such hydrogen-bonds is around 1 ps. Hydrogen-bonding is the main responsible of the amazing properties of liquid water. Water beyond its critical point (T > 647.13 K, d > 0.322 g/cm³, P > 220.55 bar) shows surprising new properties, like the capacity of mixing with oil. In supercritical water there is no longer distinction between the liquid and vapor phase. We have observed the breakdown of the tetrahedral structure and its substitution by cavities and water clusters. The lifetime of hydrogen-bonds is now about 0.3-0.5 ps. This part of my research has been carried out in collaboration with Prof. Elvira Guàrdia (UPC) and with Prof. Carmen Gordillo (University Pablo de Olavide, Seville). |
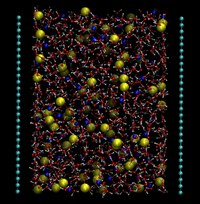
Aqueous ionic solutions at finite concentration
The effect of ion concentration is relevant to the local organization of water around ionic species and it has strong influence on dielectric properties and on dynamics, such as conductivity and diffusion coefficients or on hydrogen-bond structure and lifetimes. Our main contribution in this field has been the use and development of polarizable models for water and ions, which have greatly improved the quality of simulation results at interfaces. This work has been done in collaboration with Prof. Elvira Guàrdia (UPC) and PhD Jonas Sala (UPC).
B. Rare events in Chemical Physics and Biophysics
|
Lipid chain performing a flip-flop transition in a biological membrane |
|
|
The study of rare events is a computational challenge due to the tiny amount of trajectories in phase space which lead to such phenomena, compared with the total amount of trajectories described by a microscopical system. A new technique named transition path sampling, which is able to analyze reactive trajectories in configurational space without preconceived information about potential energy surfaces or transition states of the system, was developed by the research group of Prof. David Chandler in the University of California at Berkeley. We have employed that technique to study the transition state structure of NaCl dissociation in water and, more recently, flip-flop transitions of lipids in biomembranes. That reserach was done in collaboration with David Chandler (University of California, Berkeley) concerning NaCl dissociation and also Félix S. Csajka (PhD from U.C.Berkeley) about NaCl dissociation and lipid transitions. |
C. Liquid and supercritical water under confinement
|
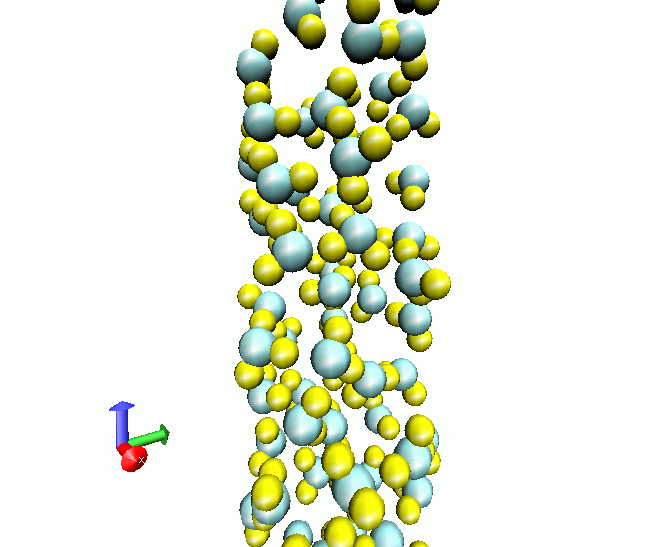
Liquid water at 298 K adsorbed in a carbon nanotube of radius 40 nm
|
 |
|
When liquids are constrained inside solid devices, its behavior can suffer dramatic changes. In the case of water, confinement produces marked loss of structural order and modifications in its microscopic dynamics. Spectroscopy is of fundamental importance (see How to interpretate the low-frequency spectrum of liquid water and a public debate). We have simulated several liquid and supercritical water samples when constrained by rigid and soft carbon nanotube walls. One of the most relevant cases is that of quasi-one dimensional liquid water, when liquid water is simply composed by linear chains constrained inside a nanotube with a radius of approximately 26 nm! The influence of the tubes is especially relevant concerning hydrogen stretching vibrations and diffussive behavior. This research work was produced in collaboration with M.C.Gordillo (Pablo de Olavide University at Seville), Dr. Carles Calero and Elvira Guàrdia (UPC). |
|
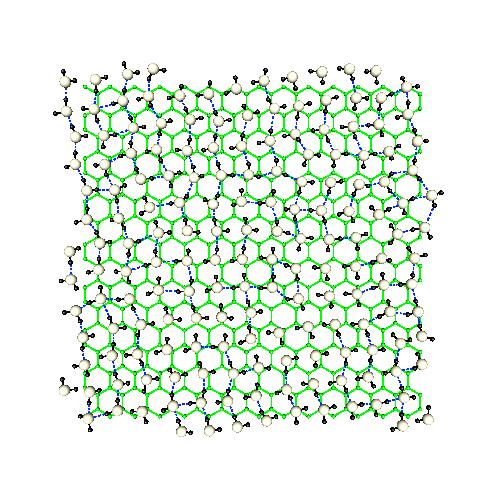
The water-solid interface
|
|
|
As another example of confinement, we have simulated liquid water imbedded in between two flat graphite layers. This is a "classical" system in simulation studies which we will use to learn further about modifications in the structure and dynamics of water under extreme confinement. In the limit, we are able to speak about 2D water.The influence of the tubes is especially relevant concerning hydrogen stretching vibrations and diffussive behavior. This research work was produced in collaboration with M.C.Gordillo (Pablo de Olavide University at Seville), Prof. Gabor Nagy (Atomic Energy Research Institute of the Hungarian Academy of Sciences) and Carles Calero and Elvira Guàrdia (UPC). |
|
 |
 |
D. Proton transfer in aqueous environments
|
In supercritical water
|
|
A study of structure and dynamics of water plus an excess proton at the supercritical 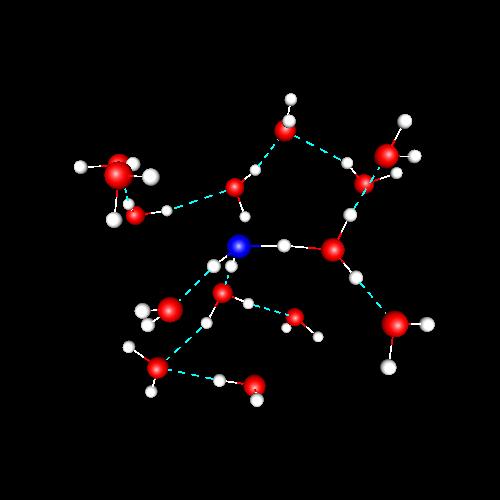
isotherm of 673 K was reported in 2004, revealing new features. For instance, the diffusion behaviour of the lone charge does not follow the Grotthus mechanism, differently of what happens for water at ambient conditions. In supercritical water, the spectral features of the excess proton reveal a tendency to form Eigen-like complexes, instead of the typical behaviour of Zundel pairs at 298 K. This part of my research was carried out in collaboration with Elvira Guàrdia (UPC) and with Prof. Daniel Laria, from the National Comission of Atomic Energy in Buenos Aires (Argentina). |
|
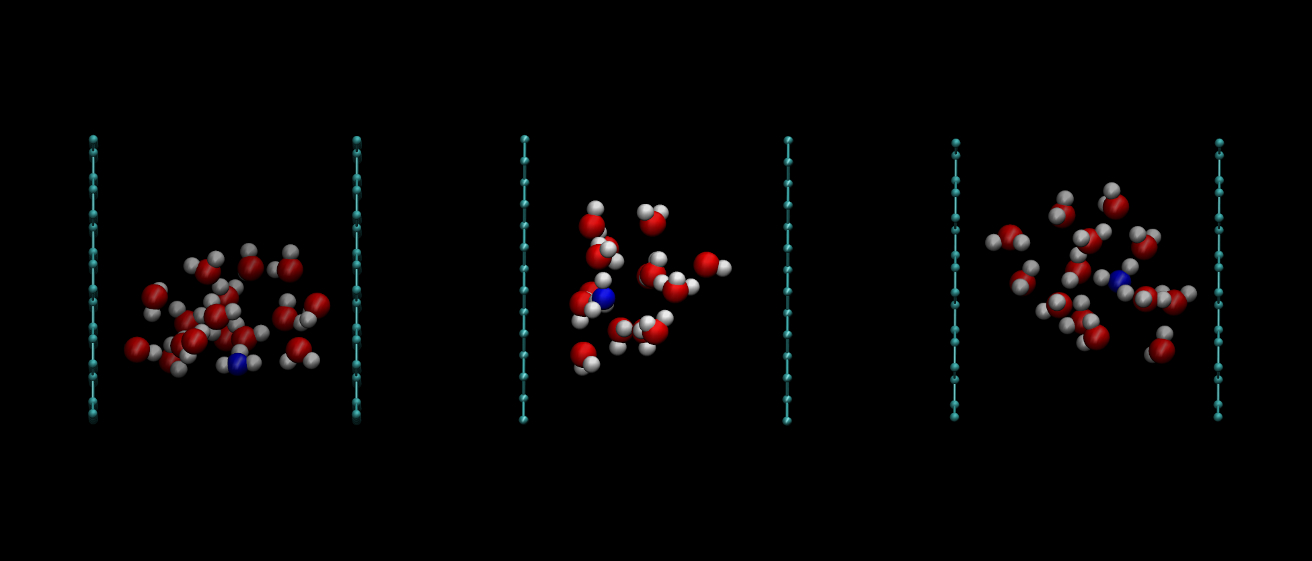
Excess protons in reverse micelles
|
||
|
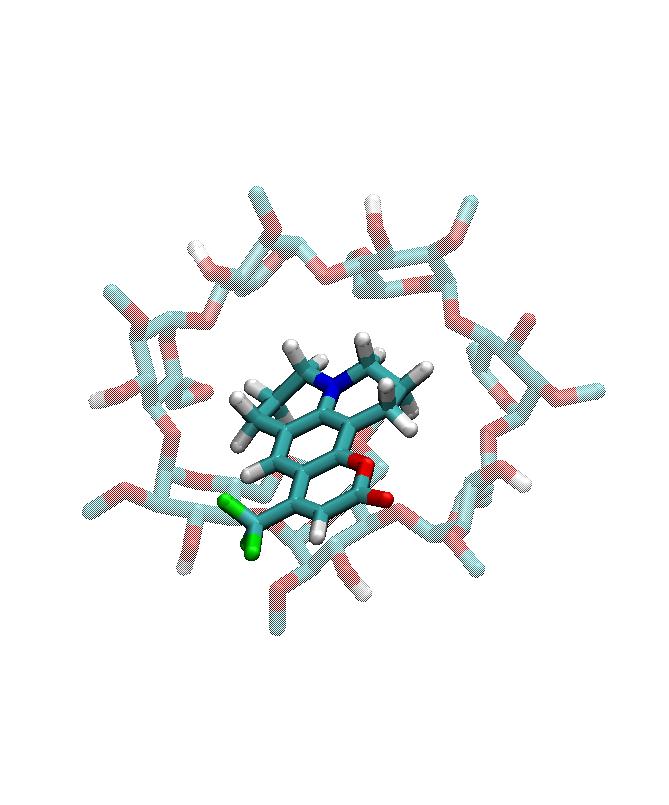
More recently, the analysis of the characteristics of the excess proton in a water droplet located inside a reverse non-ionic micelle Diethylene Glycol Monodecyl Ether has been performed. Is such system, we observe a quick diffusion of proton to the external surface of the micelle, in a similar fashion as it happens when larger ionic charges are dilutes near solid or gas-like interfaces. In this particular problem, we observed how the most common configuration is that of the excess proton forming Eigen-like complexes, which have long lifetimes of the order of nanoseconds, prior to tits eventual transfer to other water molecules. This part of my research was carried out in collaboration with Elvira Guàrdia (UPC) and with Daniel Laria and Dr. Javier Rodriguez, from the National Comission of Atomic Energy in Buenos Aires (Argentina).
|
E. Modeling and simulation of cell membranes
Back in 2013, I started a new research line based on the modeling and simulation of cell membranes, made of bilayer lipid surfactants inside an aqueous environment. We started simulating the cell membrane at all-atom level, including all sites and interactions. It means, for instance, to move 128 lipid sites of DMPC lipid molecules inside a sea of 3654 water molecules at the simplest level or considering the interaction of +200000 atomic sites for a protein inside a cell membrane. This work started as a collaboration with PhD Jing Yang, with Carles Calero (University of Barcelona) and is actually carried out together with Dr. Huixia Lu ("Margarita Salas" fellow at UPC). Our main aim is to significantly improve the realism of the membrane, including ionic species, cholesterol and selected proteins and, especially, to enlight the interactions of small molecuels (tryptophan, melatonin, serotonin), proteins and drugs within the membrane. The most recent developments made by PhD student Zheyao Hu (CSC fellow) involve the simulation of proteins KRas at cellular interfaces, simulating systems of +200000 atoms at the all-atom level and including new drugs designed in silico able to modifiy the activation of the oncogenes at the atomic level.
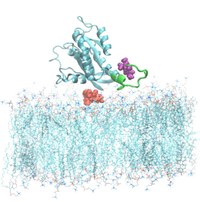
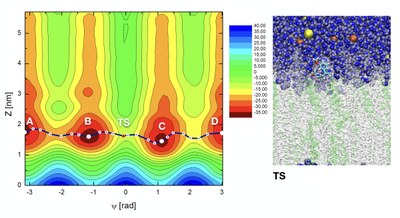
F. Helium nucleation in fusion reactor blankets

In 2019 I started a collaboration with Profs. Lluís Batet (UPC), Ferran Mazzanti (UPC) and Grigory Astrackharchik (UPC) to study the phenomenon of helium nucleation inside the lead-lithium blankets of nuclear fusion reactors. We are currently developing force fields and conducting Molecular Dynamics and Monte Carlo simulations of helium-lithium mixtures in the isothermal-isobaric ensemble, such that at 843 K and pressures between 1 and 10000 bar, helium bubbles have been observed as stable structures evolving inside the lithium solvent. The joint influence of temperature and pressure is essential for the formation and eventual stability of the bubbles. The next development will consider the eutectic mixture of lithium and lead as the embbedding solution.
G. Design and simulation of new drugs and compounds for cancer treatment
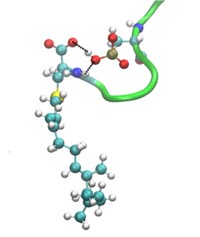
Following the research on biosystems, recently I've started a new research line based on the simulation of oncogenic proteins near cell membranes or within aqueous ionic environments. We have devoted our recent and current research work to the simulation of oncogenes of the RAS family at the all-atom level. Up to date, we considered the KRAS and NRAS sub-sets, in particular analysing their structure and dynamics using Molecular Dynamics and metadynamics simulations, in the time range of microseconds. KRAS is mainly related to the pancreatic, lung and colon cancers, whereas NRAS in directly connected to the melanoma skin cancer. Our most relevant findings include the description of the microscopic mechanism used by KRAS-4B to anchor in a cell membrane and the location of specific drugging pockets inside KRAS and NARS. In the KRAS case we have designed a prototype drug (DBD-15-21-22) that can block the oncogen in its inactive state. We have also designed a protocol method ("Isomer-sourced structure iteration") able to provide a systematics for the desing of new compounds potentially effective as drugs. In the near future we plan to study the family of MYC oncogenes in order to locate new strategies for drugging them, which are still lacking.
This work is a collaboration with PhD students Zheyao Hu and Yanhong Ge (both awarded with "China Scholarship Council' s grants) and Dr. Huixia Lu ("Margarita Salas" fellow at UPC).
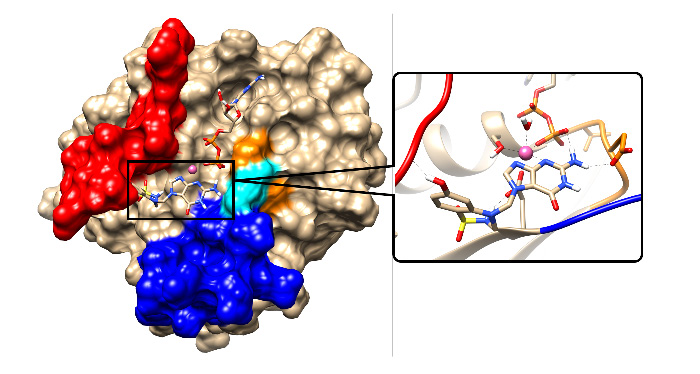
Prototype inhibitor DBD-15-21-22 (right) targetting GDP-Mg-KRAS-G12D (left).
Publication list
Books
Liquid water and aqueous solutions (bulk and at interfaces)
Proton transfer in aqueous systems
Rare events in biochemical systems. Cell membranes
Scientific parameters (Google Scholar, December 2025)
Index h: 40
Index i10: 73
Citations: 4425
ORCID:

Teaching activities
A. PhD students
Actually I'm the thesis (co)advisor of three PhD students:
B. Courses
- Advanced Methods in Simulation, elective topic for students of the interuniversitary Master of Science on Atomistic and Multiscale Computational Modelling in Physics, Chemistry and Biochemistry (UB-UPC)
-
Physics, for Computer Science students, compulsory topic. Faculty of Computer Science, UPC, Barcelona.
- Computer Simulation in Condensed Matter, elective topic at the degree of Physics Engineering at UPC, Barcelona
-
Physics of Memory Devices, elective topic at the degree of Computer Science at the Faculty of Computer Science, UPC, Barcelona.
Academic governing activities
1. Member of the Academic Comission for the Evaluation of Professors (Department of Physics, UPC).
2. Head of the Physics Section at the UPC's Physics Department at the Computer Science School and the Telecommunications School (UPC).

Share: